
Five components.
autologIX concentrates the complete biological content of plasma — not just platelets. Five therapeutic components are retained and concentrated through the ProtSmart 6 ultrafiltration membrane (15 kDa molecular weight cutoff).
CHARACTERIZED EXOSOMES AND EXTRACELLULAR VESICLES
Analytical testing measured approximately 30 trillion particles per milliliter. Flow cytometry confirmed expression of CD9 (93.2%), CD63 (90.1%), and CD81 (69.1%) — the three canonical EV markers recognized by the MISEV2023 international standard. TEM confirmed vesicular morphology. All EVs (30–1,000 nm) are physically retained by the membrane’s 5 nm pores.
Source: StemExoOne Co., Ltd., 2024 (commissioned analytical testing).
CONCENTRATED PLASMA PROTEINS
The 15 kDa membrane retains all plasma proteins above its molecular weight cutoff: fibrinogen (340 kDa) for fibrin scaffold formation, alpha-2-macroglobulin (720 kDa) for protease inhibition, fibronectin (440 kDa) for cell adhesion, and albumin (66.5 kDa) for growth factor transport. Published studies report approximately 2× protein enrichment using ultrafiltration (Mazzucco et al. 2024, Frontiers in Bioengineering and Biotechnology).
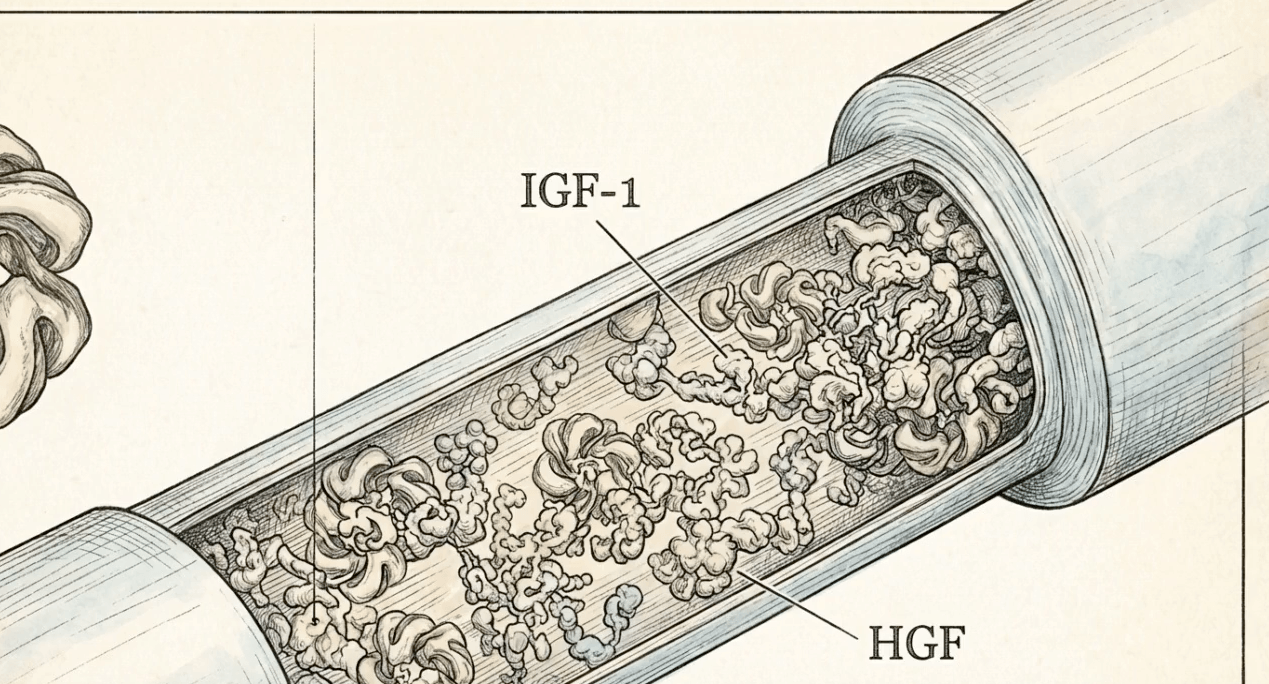
DUAL-AXIS GROWTH FACTORS
Platelet-derived growth factors (PDGF, VEGF, TGF-β, EGF) are concentrated alongside extraplatelet factors (IGF-1, HGF) that standard centrifugation PRP cannot enrich. Published data show IGF-1 is the only growth factor correlated with cell proliferation, and it resides in plasma rather than platelets (Beitia et al. 2023, International Journal of Molecular Sciences).
PLATELETS
Platelets are retained above the gel barrier during centrifugation and concentrated during ultrafiltration. The larger output volume (approximately 6–7 mL) is designed to deliver a higher total platelet dose to the treatment site.
FIBRIN SCAFFOLD PRECURSORS
Concentrated fibrinogen, upon activation, forms a dense autologous fibrin matrix. Published research shows fibrinogen binds at least 15 growth factors with nanomolar affinity, creating a sustained-release reservoir lasting up to 7 days (Martino et al. 2013, PNAS).
Analytical testing measured approximately 30 trillion particles per milliliter. Flow cytometry confirmed expression of CD9 (93.2%), CD63 (90.1%), and CD81 (69.1%) — the three canonical EV markers recognized by the MISEV2023 international standard. TEM confirmed vesicular morphology. All EVs (30–1,000 nm) are physically retained by the membrane’s 5 nm pores.
Source: StemExoOne Co., Ltd., 2024 (commissioned analytical testing).
Exosome and
The following analytical data characterize the extracellular vesicle content of the autologIX concentrate. These results are from commissioned laboratory testing aligned with the three core pillars of MISEV2023, the international standard for EV characterization contributed to by over 1,000 scientists. They have not been published in peer-reviewed literature.
Note: These analytical results characterize the concentrate’s composition. They do not constitute clinical evidence of therapeutic efficacy. Clinical studies investigating autologIX outcomes are planned.
Approximately 30 trillion particles per milliliter. Size distribution consistent with extracellular vesicle populations (median size: 154.7 nm).
Source: StemExoOne Co., Ltd., 2024.
30T/mL Exosomes
Secures approximately 30 trillion exosomes per mL from autologous blood
Nanoparticle tracking analysis (NTA)
[Reference] 1. StemExoOne Co., Ltd., Independent Validation Report, 2024
Uniform Exosome Size
Confirmed uniform size and structural consistency
Distribution of Particles
Surface marker analysis confirmed expression of three canonical EV tetraspanins: CD9 (93.2% positive), CD63 (90.1% positive), CD81 (69.1% positive).
Source: StemExoOne Co., Ltd., 2024.
ProtSmart 6 CD Marker
Confirmed expression of effective exosome markers (CD9, CD63, CD81): proven high positive rates
Flow Cytometry
[Reference] 1. StemExoOne Co., Ltd., Independent Validation Report, 2024
Vesicular structures with morphology consistent with extracellular vesicles confirmed at nanometer resolution.
Source: StemExoOne Co., Ltd., 2024.
Transmission Electron Microscopy

Comparative multiplex analysis showed elevated concentrations of PDGF-BB, EGF, and VEGF in autologIX output versus conventional PRP preparation.
Source: Wongbee MedTech, Report 2025-027-L.5Plex.
Growth Factor
Generates up to 4.8 times more effective growth factors compared to conventional PRP
Multiplex Growth Factor Quantification (5-Plex)
- PRP
- ProtSmart 6
(Unit: pg/mL)
[Reference] 1. Wongbee MedTech Report (2025-027-L.5Plex).
What every centrifugation
Standard PRP preparation uses centrifugation to concentrate platelets — then discards 60–80% of starting plasma volume as platelet-poor plasma (PPP). With that discarded plasma go therapeutic molecules that centrifugation cannot concentrate.
Published data demonstrate:
IGF-1
The only growth factor independently correlated with cell proliferation (Beitia et al. 2023, IJMS). Standard PRP shows zero enrichment versus unprocessed blood (p = 0.9995). Protein-concentrated PRP doubles IGF-1 (p < 0.0001). Source: Sánchez et al. 2025, Nature Scientific Reports.HGF
Hepatocyte growth factor, anti-inflammatory and anti-fibrotic. No enrichment in standard PRP (p = 0.4536). Significantly enriched by protein concentration (p < 0.0001). Source: Sánchez et al. 2025.Alpha-2-macroglobulin (A2M)
The master protease inhibitor of cartilage-degrading enzymes (Wang et al. 2014, Arthritis and Rheumatology). Concentrated approximately 2-fold by protein enrichment (p < 0.0001). Source: Sánchez et al. 2025.Extracellular vesicles
Protein-concentrated PRP formulations contain significantly higher EV concentrations (1.57-fold increase, p = 0.019). Source: Sánchez et al. 2025.Why centrifugation cannot solve this: Centrifugation separates particles by density. Dissolved plasma proteins share the same density as the plasma carrier — approximately 1.025 g/mL. No centrifuge speed, duration, or blood volume can concentrate a dissolved molecule. Ultrafiltration separates by molecular size through a 15 kDa membrane — the only point-of-care method that concentrates dissolved plasma proteins.
How autologIX
STEP 1: GEL SEPARATION (SINGLE SPIN)
Blood is drawn into four Swiss Platelet Gel Life (SPGL) tubes (12 mL each, 48 mL total). After centrifugation per IFU parameters, the separator gel retains red cells and granulocytes below while platelets and plasma proteins stratify above the gel barrier.
STEP 2: PROTSMART 6 ULTRAFILTRATION
Recovered plasma is processed through the ProtSmart 6 hollow-fiber ultrafiltration device (15 kDa molecular weight cutoff). The membrane removes plasma water, salts, and low-molecular-weight solutes while retaining platelets, plasma proteins, growth factors, and extracellular vesicles. No additional centrifugation is required for concentration.
DRAW
48 mL whole blood into 4× SPGL tubes (12 mL each).
CENTRIFUGE
Single spin per IFU parameters. Gel separator retains red cells; platelets and proteins stratify above.
ULTRAFILTER
ProtSmart 6 concentrates to target volume. Plasma water exits through the 15 kDa membrane; everything therapeutic stays.
RECOVER
Approximately 6–7 mL protein-enriched concentrate. Use as injectable PRP, apply topically, or activate to form an autologous fibrin scaffold.
SYSTEM AT A GLANCE:
48 mL — Blood draw
4 tubes — SPGL configuration
15 kDa — Membrane cutoff
≈ 20 min — Total procedure
≈ 6–7 mL — Output volume
ProtSmart 6
At the core of autologIX is the ProtSmart 6 ultrafiltration device — a precision hollow-fiber system manufactured by Medica S.p.A. (Medolla, Italy) that separates plasma components by molecular weight.
800 Tubes


This is a zero-loss concentration principle: unlike additional centrifugation spins that risk losing therapeutic content, ultrafiltration removes only non-therapeutic components.
Technology validation in published research
The ProtSmart filter platform (same manufacturer, Medica S.p.A.) has been used in published clinical research. Mazzucco et al. (2024) used ProtSmart-based ultrafiltration to prepare protein-enriched filtered PRP for treating Stage III/IV pressure ulcers in a 10-patient series, demonstrating 52% wound size reduction at 6 weeks and identifying 30 upregulated proteins across 8 functional categories. Zero adverse events were reported. (Frontiers in Bioengineering and Biotechnology, 11:1279149.)
This is a zero-loss concentration principle: unlike additional centrifugation spins that risk losing therapeutic content, ultrafiltration removes only non-therapeutic components.
Technical Specifications:
Membrane material: MediSulfone® polysulfone
Manufacturer: Medica S.p.A. (Medolla, Italy)
Fiber count: 800 hollow fibers
Molecular weight cutoff: 15 kDa
Pore diameter: ≤ 5 nm
UF processing time: approximately 8 minutes
Input: recovered plasma from 4 × 12 mL SPGL
Published evidence
While direct clinical studies with autologIX are in development, the ultrafiltration PRP approach is supported by a growing body of peer-reviewed literature from independent research groups.
TECHNOLOGY VALIDATION (ProtSmart filter platform)
Technology validation in published research
The ProtSmart filter platform (same manufacturer, Medica S.p.A.) has been used in published clinical research. Mazzucco et al. (2024) used ProtSmart-based ultrafiltration to prepare protein-enriched filtered PRP for treating Stage III/IV pressure ulcers in a 10-patient series, demonstrating 52% wound size reduction at 6 weeks and identifying 30 upregulated proteins across 8 functional categories. Zero adverse events were reported. (Frontiers in Bioengineering and Biotechnology, 11:1279149.)
This is a zero-loss concentration principle: unlike additional centrifugation spins that risk losing therapeutic content, ultrafiltration removes only non-therapeutic components.
PROTEIN AND EV ENRICHMENT
Sánchez et al. (2025) demonstrated that PRP with elevated protein content showed significantly higher EV concentrations (p = 0.019), enhanced cell proliferation (p < 0.001), and decreased pro-inflammatory cytokines from M1 macrophages. Nature Scientific Reports, 15:4523.
Mercader Ruiz et al. (2023) validated that ultrafiltration concentrates extraplatelet growth factors including IGF-1 (p < 0.01) and HGF (p < 0.001) while preserving ionic balance and pH. Journal of Clinical Medicine, 12(18):5941.
Everts et al. (2024) characterized protein-rich PRP as a multi-purpose biological platform where concentrated fibrinogen creates denser fibrin scaffolds with sustained growth factor release. International Journal of Molecular Sciences, 25(14):7914.
IGF-1: THE MISSING VARIABLE
Beitia et al. (2023) demonstrated that IGF-1 is the only growth factor correlated with cell proliferation in PRP — and it does not correlate with platelet count, confirming its plasma origin. This is a factor that conventional centrifugation PRP cannot concentrate. International Journal of Molecular Sciences, 24(6):5367.
DOSE-RESPONSE EVIDENCE
Berrigan et al. (2025) found in a systematic review of 29 RCTs that positive PRP outcomes required mean platelet doses above 5 billion (p < 0.01). Arthroscopy, 41(3):809-817.
Patel et al. (2024) demonstrated in a Level 1 triple-blind RCT that 8 mL PRP significantly outperformed 4 mL for knee OA outcomes at 6 months (p < 0.001), with 96% patient satisfaction versus 68%. Orthopaedic Journal of Sports Medicine.
How the concentrate
The biological activity of autologIX's protein-enriched concentrate involves an integrated set of processes described in published literature.
PLATELET AND EV SIGNALING
Upon administration, activated platelets and their extracellular vesicles deliver regulatory cargo — growth factors, microRNAs, and signaling proteins — to recipient cells. Published data show these signals promote cell proliferation, angiogenesis, and collagen synthesis (Sánchez et al. 2025).GROWTH FACTOR COOPERATION
Concentrated platelet-derived growth factors (PDGF, VEGF, TGF-β, EGF) combined with extraplatelet factors (IGF-1, HGF) concentrated via ultrafiltration create a coordinated biological signal for tissue repair (Everts et al. 2024; Mercader Ruiz et al. 2023).INFLAMMATION MODULATION
Published in vitro data demonstrate that protein-enriched PRP formulations significantly decrease pro-inflammatory cytokines (IL-1β, IL-6, TNF-α) in M1-type macrophages, promoting transition from inflammatory to reparative tissue states (Sánchez et al. 2025).SCAFFOLD FORMATION
Elevated fibrinogen in the concentrate, upon activation, forms a dense autologous fibrin matrix. This matrix provides structural support, sustained growth factor release for up to 7 days, and a microenvironment for cell migration and tissue remodeling (Everts et al. 2024; Martino et al. 2013, PNAS).Areas of
autologIX's higher-volume output (approximately 6–7 mL) is designed for applications where standard PRP volumes may be insufficient. Use is at clinician discretion and subject to local regulations.
ORTHOPEDICS
9 in 10 positive knee OA trials delivered >5 billion platelets per injection (Berrigan 2025)
WOUND CARE
52% wound area reduction at 6 weeks with the same ProtSmart filter platform (Mazzucco 2024)
HAIR RESTORATION
IGF-1: the only growth factor correlated with follicle proliferation — standard PRP misses it
SPORTS MEDICINE
PRP delivers significantly better long-term outcomes than corticosteroids for tendinopathies
SEXUAL WELLNESS
First placebo-controlled RCT: 69% achieved meaningful improvement versus 27% placebo
GYNECOLOGY
43-study scoping review: PRP improved symptoms for lichen sclerosus and vulvovaginal atrophy
Explore the autologIX
Product and Technology



Clinical Fields
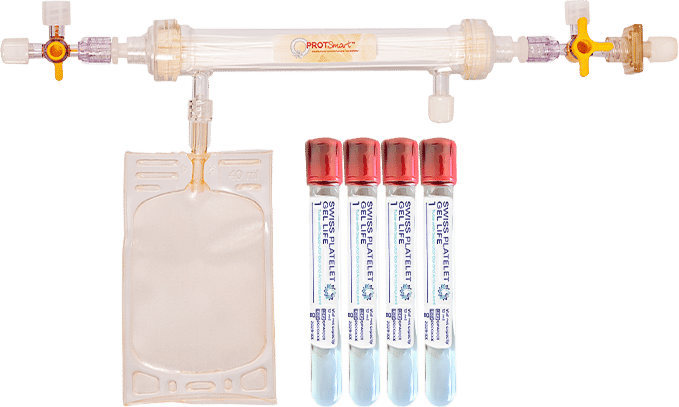
Kit and
Kit contents:
- 4× Swiss Platelet Gel Life (SPGL) tubes with separator gel (12 mL each)
- ProtSmart 6 ultrafiltration device
- blood-draw accessories (by configuration)
- Sterile syringes
- Quick protocol card
- Instructions for use.
Quality and documentation: Defined centrifugation parameters (time/RCF/volumes per IFU), ultrafiltration steps and target volumes, SOPs, batch/QC templates, and traceability documentation. CE-marked where applicable; availability and indications vary by market. For professional use only.
Key
CE-MARKED COMPONENTS
Medical device components carry CE marking where applicable100% AUTOLOGOUS
Uses only the patient's own blood; no additives or donor materialsCHARACTERIZED EXOSOMES
Exosome content confirmed by NTA, flow cytometry, and TEMDUAL CONCENTRATION
Platelets and plasma proteins concentrated in a single workflowREPRODUCIBLE
Membrane-based concentration reduces operator variabilityDEFINED PARAMETERS
Standardized centrifugation and ultrafiltration per IFUHIGHER VOLUME
6–7 mL output for applications requiring larger injectate volumesSafety
Contraindications:
autologIX should not be used in patients with: active systemic infection or bacteremia; platelet dysfunction syndromes or critical thrombocytopenia (platelets below 100,000/μL); hemodynamic instability; known hypersensitivity to sodium citrate anticoagulant; hemoglobin below 10 g/dL; hematologic malignancy. Malignancy at the treatment site is a relative contraindication at clinician discretion.
Warnings:
PRP preparation involves standard venipuncture risks including hematoma, phlebitis, and vasovagal reaction. Sterile technique must be maintained throughout preparation and administration. The final concentrate is intended for same-day use only and must not be stored. All kit components are single-use only. Do not resterilize.
Important:
autologIX is a medical device for the preparation of autologous platelet-rich plasma. It is not a drug or biologic. PRP therapy outcomes vary by patient, indication, and preparation method. Clinicians should exercise independent medical judgment.
Frequently asked
autologIX by SWISS concept is a medical device system for preparing protein-enriched autologous platelet-rich plasma from a patient’s own blood. It uses gel separation and ProtSmart 6 ultrafiltration to concentrate platelets, plasma proteins, growth factors, and extracellular vesicles into approximately 6–7 mL of output from a 48 mL blood draw.
Yes. Analytical testing using three methods — nanoparticle tracking analysis (30 trillion particles/mL), flow cytometry (CD9 93.2%, CD63 90.1%, CD81 69.1%), and transmission electron microscopy — confirmed the presence of extracellular vesicles with exosome-associated markers. These three methods align with the core pillars of MISEV2023, the international standard for EV characterization.
Most PRP systems use centrifugation alone, which concentrates platelets but discards 60–80% of plasma volume along with its dissolved therapeutic proteins. autologIX uses ultrafiltration through a 15 kDa membrane that removes only water and small solutes, retaining platelets and plasma proteins together in a single concentrate.
Approximately 20 minutes from blood draw to ready-to-use concentrate.
autologIX requires a standard laboratory centrifuge for the gel separation step. The ProtSmart 6 ultrafiltration device is included in the disposable kit. No specialized PRP centrifuge or automated system is required.
The ultrafiltration approach is supported by peer-reviewed literature from independent research groups including Mazzucco et al. (2024), Sánchez et al. (2025), Mercader Ruiz et al. (2023), and Everts et al. (2024). The same ProtSmart filter platform (Medica S.p.A.) was used in the Mazzucco clinical study. Direct clinical studies with autologIX are in development.
